Scientists show how cancer cells repair DNA
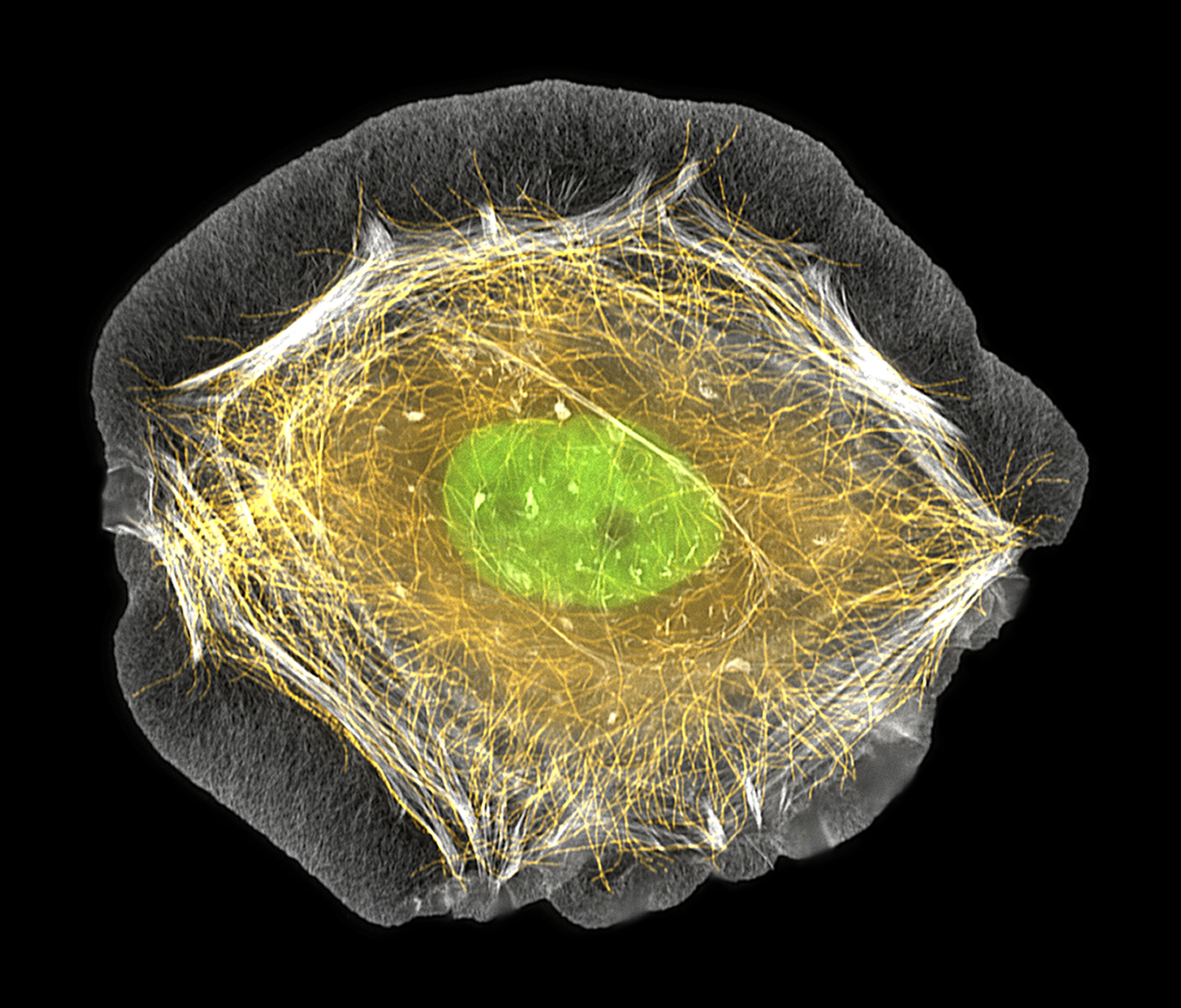
Researchers at the University of Geneva have discovered a mechanism, which allows human tumour cells to repair damaged genetic structures necessary to complete their division, a finding which may lead to new approaches to fight cancer.
Thanos Halazonetis from the University of Geneva and colleagues in Finland, Germany, the United States and Sweden identified a repair process that had previously been observed in yeast but not in humans, according to a study published online by the journal Science on Thursday.
For cells to divide, they must replicate their DNA which consists of around 6.4 billion pairs of nucleotides, the building blocks of nucleic acids. The double-stranded DNA opens up like a zipper, producing a so-called replication fork. It has been known that cancer cells have damaged forks and that they manage to repair them, but the mechanism was not clear.
“The most important finding is that we now know which pathway cancer cells use to repair broken replication forks so they can resume replication,” Halazonetis told swissinfo.ch.
In the development of cancer, specific genes known as proto-oncogenes control cell proliferation. Their mutation into oncogenes triggers an uncontrolled cell growth and promotes the development and spread of cancer.
In tumour cells, these oncogenes induce a collapse or even a rupture of the replication forks, explained Halazonetis. This mechanism is aimed at avoiding the proliferation of cancerous cells, but the ability to self-repair the forks can overcome this.
The geneticists analysed 690 genes involved in DNA metabolism and built up a library of molecules capable of preventing gene regulation.
The researchers were then able to isolate several genes essential for the repair of damaged forks. It allowed them to identify a repair process called break-induced replication, which is very common in cancer but rare in healthy cells.
Halazonetis’ next objective is to identify all the other biochemical players involved in this mechanism to determine which ones could be used as therapeutic targets.
“Our main goal now is to inhibit this pathway,” Halazonetis said. “I have no doubt that further research will yield new targets, whose activity may be modified by a drug, but we are never sure whether if they will translate into new therapies.”

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative
You can find an overview of ongoing debates with our journalists here. Please join us!
If you want to start a conversation about a topic raised in this article or want to report factual errors, email us at english@swissinfo.ch.