Ebola vaccine trials to resume in Geneva

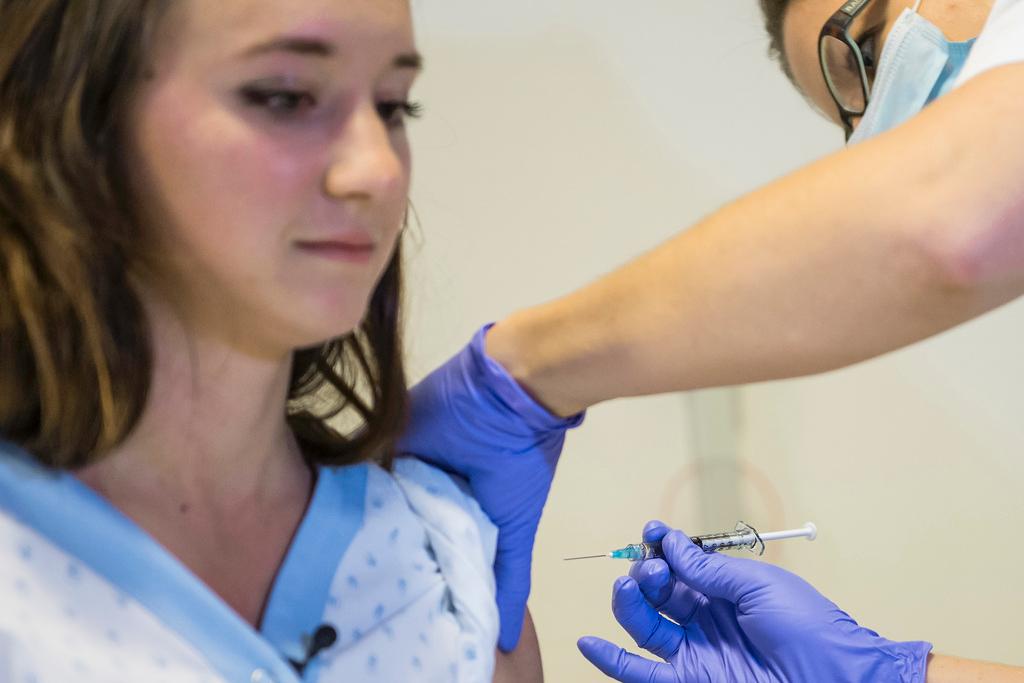
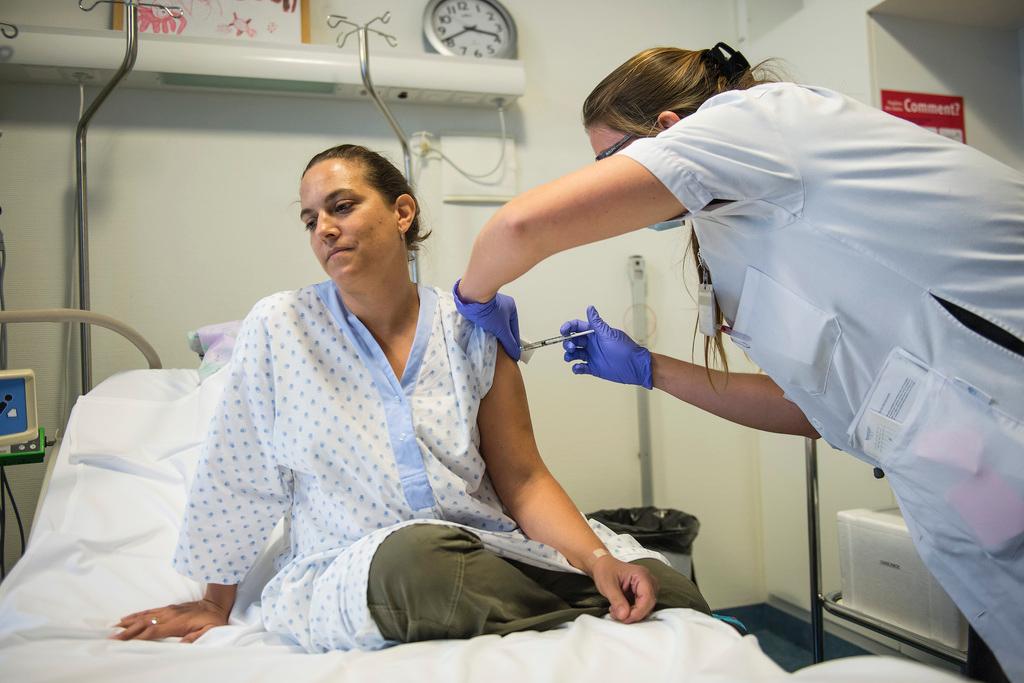
Researchers at Geneva University Hospital will resume testing one of the leading Ebola vaccine candidates on Monday, according to the hospital where part of the trial is taking place.
The tests had been suspended on December 11 as a precautionary measure after some volunteers reported unexpected side effects. The trial was scheduled to resume in January.
The study researchers had reported four cases of mild joint pain in the hands and feet of people who had received the shot ten to 15 days earlier.
The study began in November 2014. The vaccine is also being tested in the United States, Canada, Germany and Gabon. The first results are expected in March 2015, according to the Geneva Hospital.
Of the 115 people participating in the trial, 56 still need to receive the vaccine, hospital spokeswoman Laurent Kaiser told Swiss public television, RTS. The researchers will use a lower dose of the vaccine, which should be better tolerated.
The vaccine was developed by Canada’s public health agency and is licensed to two US companies, NewLink and Merck. The product, which has previously been tested on monkeys, now needs to be tested on humans.
Scientists racing to develop vaccines against Ebola are trying to determine whether they can best fight the disease with a single injection or with two, a calculation that could determine how quickly and effectively a programme can be rolled out.
The World Health Organization estimates that roughly 8,000 people have died from the Ebola outbreak in West Africa that began about a year ago. The epidemic has been centred on the countries of Sierra Leone, Liberia and Guinea.

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative
You can find an overview of ongoing debates with our journalists here . Please join us!
If you want to start a conversation about a topic raised in this article or want to report factual errors, email us at english@swissinfo.ch.