
New antibiotic offers hope against “superbug”
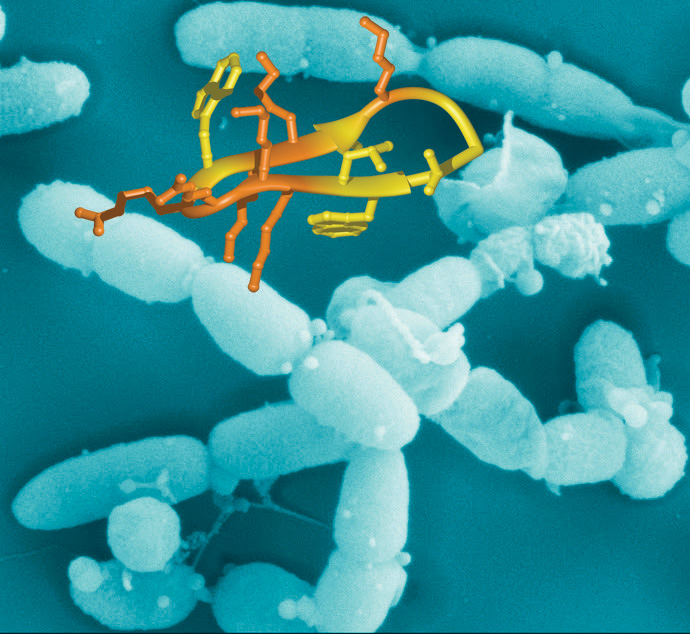
Swiss scientists have found a new class of antibiotics which target a so-called hospital “superbug” – the multi drug-resistant and often deadly pseudomonas.
They hope that mechanism by which it works – which has never been seen before – could eventually be applied to other such bugs. Big pharmaceutical companies have already expressed an interest.
The joint research team from Zurich University and Swiss biotech company Polyphor has described the new drug class as a rare and significant step in antibiotics development.
Pseudomonas aeruginosa – what is known as a “gram-negative” bacterium – is one of the most common bugs found in hospitals. One feature of gram-negative bacteria is that the structure of their outer membrane protects them from many antibiotics.
Such bacteria account for an estimated 63 per cent of infections in hospital intensive care units. Experts have voiced fears that their resistance to antibiotics is rising.
“Pseudomonas is a severe problem for immune-compromised patients, such as those with severe burns, or lung infections like pneumonia, or people who are on ventilators,” team leader John Robinson, a professor of chemistry at Zurich University, told swissinfo.ch.
It also affects those suffering from the genetic disease cystic fibrosis, where the lungs are clogged with mucus and are unable to clear bacterial infections easily.
“Eventually many of them die, not from the disease, but from the pseudomonas infection,” Robinson said.
New mechanism
There is thus an urgent need for new drugs in the fight against this type of bacteria. What makes the Swiss team’s development exciting is that it works in an entirely different way from conventional antibiotics.
Pseudomonas has a hard outer cell wall, making it difficult for a conventional antibiotic to penetrate it. What is more, if the antibiotic does manage the breach the cell’s defences, the bacterium uses a pump action to get rid of it.
“Our antibiotic actually hits a protein which is in the outer cell membrane, so it’s a sort of battering ram direct onto this essential protein machinery in the outer membrane, which is responsible for building the outer membrane,” explained Robinson.
“So we think that the antibiotic doesn’t have to get into the cell to act.”
Robinson said that it was quite rare for a completely new mechanism to be found in antibiotic research – it is believed that such events only happen every 20 years.
The results of the Swiss research were published in the February 19 edition of the renowned Science journal.
High interest
The clinical development of antibiotic is already being prepared, including Phase 1 clinical trials on healthy human volunteers.
“They are scheduled to start towards the end of the second quarter, so in summer this year,” Michael Altorfer, spokesman for Polyphor, which is based at Allschwil near Basel, told swissinfo.ch.
He confirmed that the company was already in talks with big pharmaceutical companies about developing the product, but for confidentiality reasons could not disclose any names at the moment.
“But the fact that already at this early stage big pharma partners are interested in negotiations shows that this discovery has been recognised, and the potential of these kind of molecules to be very valuable tools in the battle against this bacteria has been recognised,” Altorfer said.
However, it is likely to take some five to eight years for the antibiotic to come onto the market. It is hard to give a precise timescale, as this will depend on which patient group is targeted in Phase 2 trials – a decision most probably to be made by the pharma company developing the product, Altorfer explained.
Wider use
The antibiotic could eventually be administered in hospitals, the research team believes. Possible applications could be an inhaled form for cystic fibrosis patients, so that it would reach the lungs directly.
Robinson says that once more is understood about the new mechanism, it would in principle be possible to develop new compounds that could target other superbug bacteria.
This could include E.coli, which is also gram-negative, or the multi-resistant Acinetobacter baumannii which has been recorded in returning Iraq soldiers in both Britain and the United States and has proved difficult to treat.
But the antibiotic’s potential for treating pseudomonas is already clear. “It could be a significant step forward,” Robinson said.
Isobel Leybold-Johnson, swissinfo.ch
The team used Protein Epitope Mimetics (PEM) Technology to identify new antibiotics that either act against a broad spectrum of bacteria or selectively target one particular strain. PEM are fully synthetic, medium-size cyclopeptide-like molecules and are a class of compounds. Cyclopeptides are currently considered very interesting in antibiotic research.
The work resulted in the discovery of this new drug target and mechanism of action which kills the pseudomonas bacteria.
“The discovery of a new class of antibiotics with a novel mode of action exemplifies the power of the PEM technology to yield a new class of drugs that complements established drug classes, such as small molecules and biopharmaceuticals,” Jean-Pierre Obrecht, Polyphor’s CEO, commented in a statement.
PEM is in between these two existing classes and its molecules combine many of their advantages. In particular, they can interfere highly selectively with protein-protein interactions.
PEM is also being applied in various other fields, such as oncology and lung infections.

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative




























You can find an overview of ongoing debates with our journalists here . Please join us!
If you want to start a conversation about a topic raised in this article or want to report factual errors, email us at english@swissinfo.ch.