Ebola vaccine shows ‘promising’ results in trials
Preliminary results of tests on an Ebola vaccine trialled in Guinea have shown promising results, according to researchers at the University of Bern. They were part of an international group testing the vaccine known as rVSV-ZEBOV.
The report on the clinical trial in the West African country, which analysed 7,651 individuals, more than 3,500 of whom were vaccinated, was published in the British medical journal The LancetExternal link on Friday. It said the vaccine “might be safe and highly efficacious in preventing Ebola virus disease”.
There is currently no licensed treatment or vaccine for Ebola, which has killed more than 11,000 people in Guinea, Sierra Leone and Liberia since the world’s biggest outbreak began last year.
World Health Organization (WHO) Director-General Margaret Chan said the early results of the vaccine were “exciting and promising”. If proven effective, the vaccine could be “a game-changer”, she added.
Matthias Egger from the Institute of Social and Preventive Medicine at the University of Bern, who was involved in the trial, said: “This could finally be the beginning of the end of the Ebola epidemic in West Africa and also be useful when combating this disease in the future.”
The Ebola epidemic in West Africa has not been defeated yet, although the number of cases has dropped substantially since the start of the year.
Race to stop virus
Scientists, doctors, donors and drug companies collaborated to push the vaccine through a process that usually takes more than a decade in just 12 months.
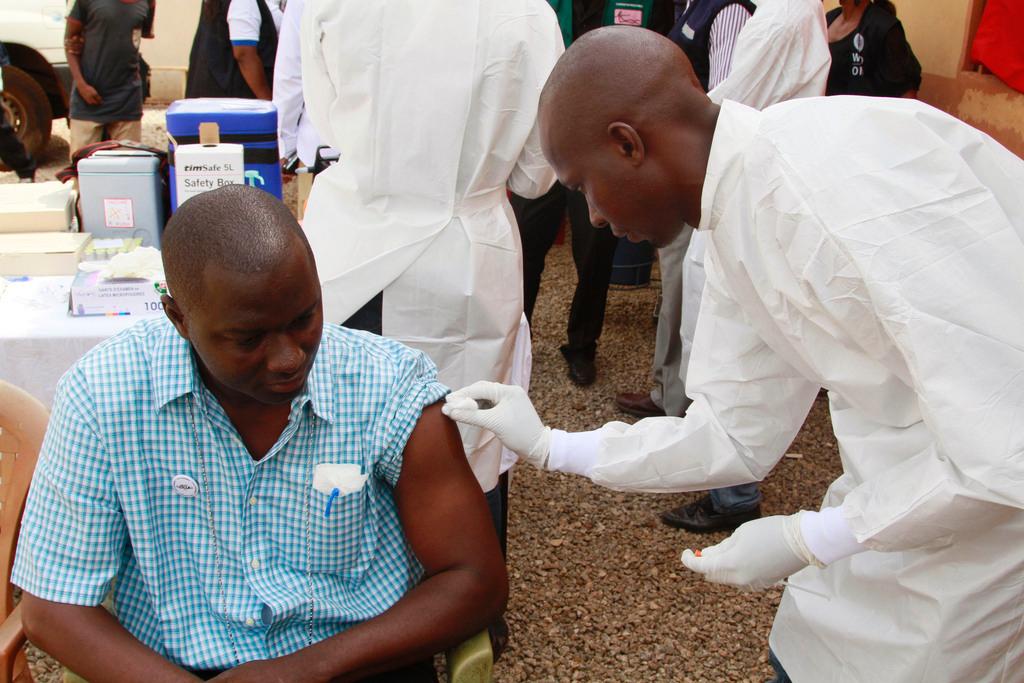
The researchers used a multi-stage approach to test the efficacy of the vaccine. Their strategy was based on “ring vaccination”, which was used to eradicate smallpox in Africa in the 1970s. People who had come into close contact with someone recently infected, and who were therefore at particularly high risk, were vaccinated.
“We can therefore say that the vaccine offers 100% protection against Ebola after roughly one week,” said Sven Trelle from the University of Bern’s Clinical Trials Unit.
Trial data will now go to regulatory agencies in the hope of getting a licence for the vaccine. This will allow it to be stockpiled for future Ebola epidemics. It is likely to be used only for people at risk in outbreaks and not given to whole populations.
WHO reform
On Friday, Chan announced that the WHO was working to reform itself in the wake of its widely criticised response to last year’s Ebola outbreak.
She said new protocols were being developed for how the agency functions during health emergencies. The agency has previously proposed creating a specialised centre within WHO to handle emergencies that would be self-managed.
Last month a report by an independent panel, commissioned to assess the WHO’s Ebola response, failed to identify a single person or even department at WHO that was responsible for the agency’s failed efforts.

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative


You can find an overview of ongoing debates with our journalists here . Please join us!
If you want to start a conversation about a topic raised in this article or want to report factual errors, email us at english@swissinfo.ch.