Swiss ready to roll out flu vaccine programme
Vaccinations against the H1N1 flu virus in Switzerland will be carried out in two phases beginning in mid-November, health authorities said on Friday.
The Federal Health Office recommends inoculating certain risk groups in a first wave, including the chronically-ill, pregnant women and health staff, but stops short of mandatory jabs. Two brands of vaccine have won the regulator’s approval.
“I hope the consensus struck between the different players involved allows us to avoid a cacophony like in many other countries,” said Thomas Zeltner, head of the Health Office.
He said an immunisation campaign could begin within the next fortnight now that the regulator Swissmedic has given the green light for two vaccines produced by the pharmaceutical companies Novartis and GlaxoSmithKline. A third brand is still being tested.
They all contain so called adjuvants, chemical compounds that increase the human body’s immune response.
Zeltner said this was in line with recommendations by the World Health Organization (WHO) and would allow Switzerland to share vaccines with less privileged countries.
He explained that without adjuvants about four times as much vaccine would be needed to achieve the desired effect.
Zeltner said Switzerland will have vaccine left following its nationwide immunisation campaign and had committed itself to support an initiative by the United States president, Barack Obama, in favour of less well-off countries.
“We are in a very privileged position and will therefore send up to 2.6 million doses of the vaccine to the WHO to assist developing countries to protect their population against flu,” he said.
The Swiss government has ordered 13 million doses of vaccine at a total cost of SFr93 million ($91 million), but patients will not have to pay for inoculations.
Two phases
The campaign, to be organised by the cantonal authorities, is due to begin by the middle of next month.
The first phase will involve up to two million of the 7.5 million Swiss population, after which the wider public will be invited to be inoculated.
“Health personnel and risk groups are the priorities,” said Franz Wyss of the cantonal health authorities.
Jacques de Haller of the Swiss Medical Association added that it was crucial that people who are in regular contact with patients in the at-risk group should get vaccinated against swine flu.
“We clearly recommend jabs for all these people even if the flu has been relatively mild in Switzerland,” he said.
He said Switzerland had good reasons to launch preparations a few months ago to brace for a major pandemic of swine flu.
“There were many deaths reported when the flu broke out in Mexico and we did not know for sure how the situation would develop,” he said.
Switzerland has recorded more than 1,550 confirmed cases of swine flu, including 30 patients who had to be admitted to hospital, but no deaths so far.
The number of people who came down with flu is believed to be higher.
Criticism
The government has come in for criticism ahead of the vaccination drive. Some doctors say the vaccines have not been sufficiently tested – a allegation rejected by the authorities.
Others question the effectiveness of the vaccine and warn of side effects.
The Health Office has also been accused of failing to communicate openly in the past few weeks and of putting off the start of the immunisation programme.
Zeltner said they had waited to present new facts at a news conference on Friday which was attended by ten representatives of the federal and cantonal authorities as well as the medical association.
New elements of an ongoing information drive, including television spots and posters, as well as an online service, will be launched in the days ahead.
Urs Geiser, swissinfo.ch

More
Pandemic
Switzerland has ordered 13 million doses of H1N1 flu vaccine from Novartis and GlaxoSmithKline.
Both vaccines contain an adjuvant called squalene, a natural organic oil which promotes good antibodies.
According to Novartis and GlaxoSmithKline, side-effects are similar to those of seasonal flu vaccines: a swelling and redness at the injection site and fever, limb pain and headache.
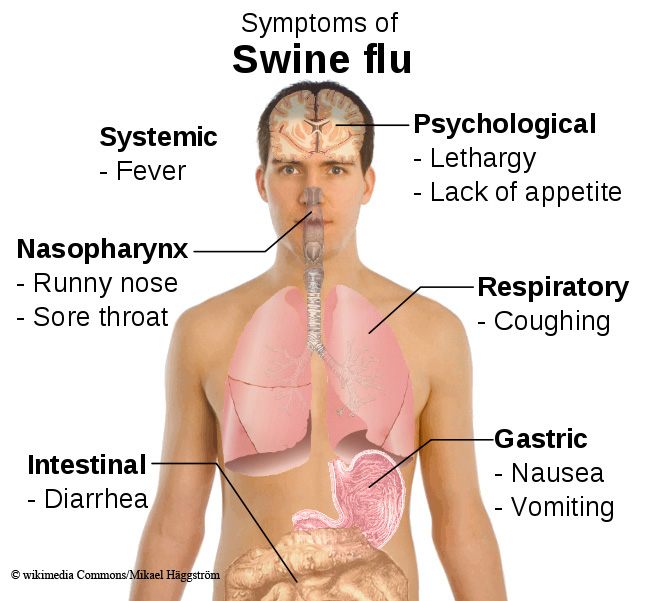
This swine flu variation, termed H1N1, is an influenza A virus spread from person to person.
It contains DNA from avian, swine and human viruses. Although swine flu viruses normally infect only pigs, they do sometimes cross the species barrier to cause disease in humans, according to the WHO.
It is passed from human to human by sneezing, coughing or by hand contact. The WHO says swine flu is not transmitted to people eating properly handled and prepared pork or other products derived from pigs.
New flu strains can spread fast because no-one has natural immunity and a vaccine can take months to develop.

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative











You can find an overview of ongoing debates with our journalists here . Please join us!
If you want to start a conversation about a topic raised in this article or want to report factual errors, email us at english@swissinfo.ch.