
Why Switzerland’s Moderna Covid-19 vaccine deal is risky
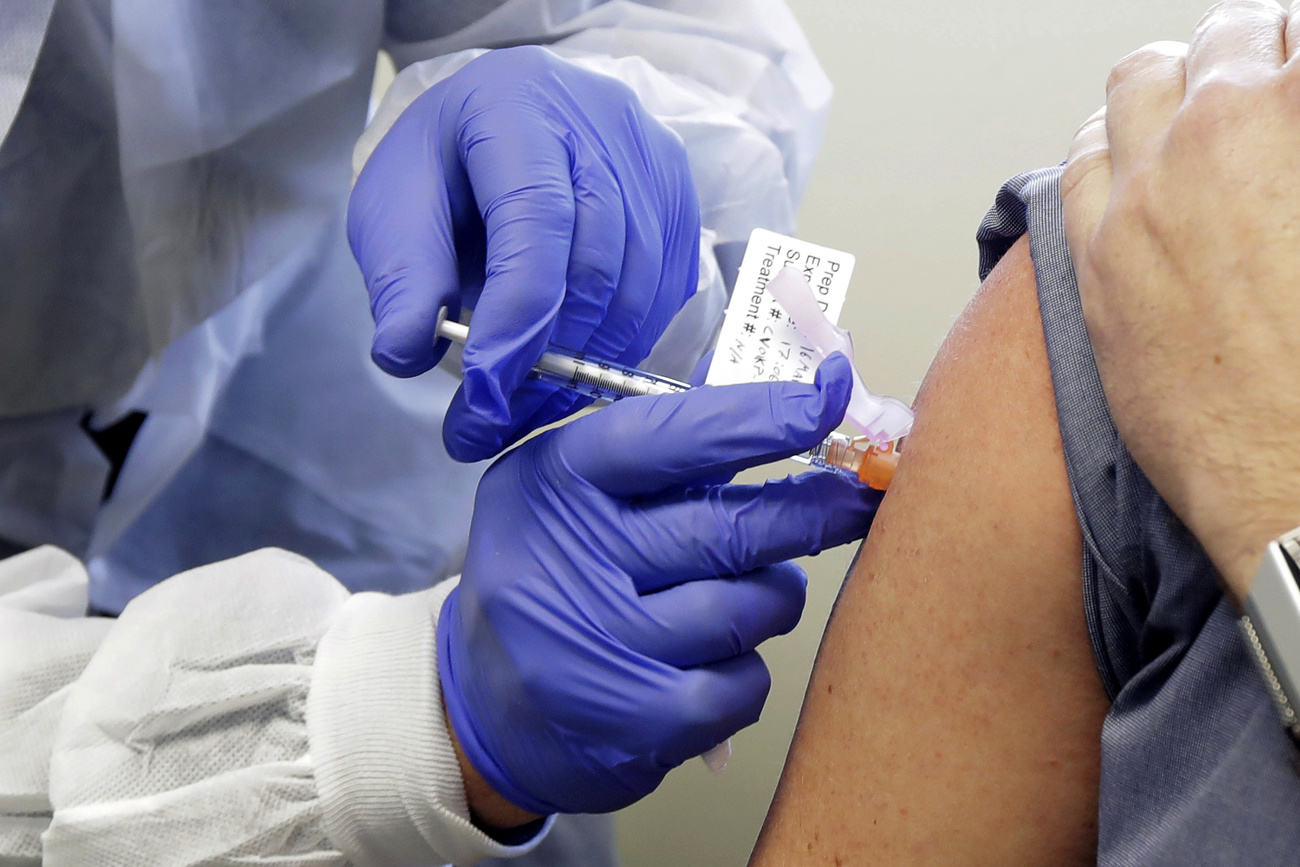
Switzerland’s order for a vaccine from American biotech firm Moderna – its first such move – is a sign that the country holds little hope for fair distribution of a Covid-19 vaccine, experts say.

With the deal signed last week, Switzerland became one of the first countries to conclude an agreement with Moderna. But it isn’t alone in signing bilateral deals with manufacturers. The United States, United Kingdom, Brazil, and Japan are among those who have already shelled out cash to pre-order promising vaccine candidates.
As the vaccine race heats up, the Moderna deal brought a sigh of relief for many in Switzerland who see it as a necessary measure to protect the Swiss population.
More
Switzerland pre-orders 4.5 million doses of Covid-19 vaccine
However, critics argue that it’s an expensive bet that ultimately undermines global efforts to ensure a fair distribution of Covid-19 vaccines – something that Switzerland has said it supports.
“This deal shows that the Swiss don’t believe a global deal to distribute vaccines fairly will be achieved,” said Patrick Durisch who leads health policy at the Swiss NGO Public Eye.
Security blanket
One of these major global efforts is the Covid-19 Global Access Facility (COVAX). Switzerland was an early backer and is now the co-chair of the global collaboration launched by the World Health Organization to speed up development and equitable access to a vaccine. It aims to procure two billion doses to vaccinate 20% of participating countries’ populations by the end of 2021.
In the announcement of the Moderna deal, the Swiss government said that it still supports multilateral projects like COVAX for the fair distribution of a future vaccine.

More
Inside Geneva podcast: Can Covid-19 ‘vaccine multilateralism’ work?
Durisch argues that the math doesn’t add up. The Economist estimatesExternal link that worldwide, governments have made future purchases of four billion doses of Covid-19 vaccines for delivery by the end of next year. This includes deals by the US and UK governments with AstraZeneca for 400 million and 100 million doses respectively. On Tuesday, the Trump administration also signed a deal with Moderna for 100 million doses.
However, the Coalition for Epidemic Preparedness Innovations (CEPI) estimates that there is only enough global manufacturing capacityExternal link to produce two to four billion doses through the end of 2021. If countries continue to sign individual deals, there simply won’t be enough for poorer countries.
So far, only AstraZeneca and Novovax have set aside doses for COVAX in addition to their bilateral deals.
But what’s the alternative? Experts predict demand will far outstrip supply for some time.
“Should we do nothing and wait to receive a vaccine?” Health minister Alain Berset asked in the German-language Neue Zürcher Zeitung newspaper over the weekendExternal link. “We are taking care of both: our own supply and a fair international distribution.”
But this gives few assurances to public health advocates like Durisch.
“It just doesn’t make sense from a public health perspective to vaccinate everyone in Switzerland but leave healthcare workers in Africa, for example, un-vaccinated,” he told swissinfo.ch. “We should be ensuring that priority risk groups such as healthcare workers receive the first vaccines regardless of where they are.”
Risky bet
The deal is also an expensive gamble. If successful, the 4.5 million vaccine doses are expected to vaccinate 2.25 million people – about a quarter of the Swiss population. Earlier this year, the Swiss government had set aside CHF300 million for vaccines and aims to procure enough to vaccinate 60% of the population.
While details are scant, the German-language Tages-Anzeiger newspaper estimatesExternal link that the Swiss government paid around CHF100-150 million ($110-160 million) to Moderna based on the biotech company’s own pricing of $32-37 a dose.
The Swiss Federal Office of Public Health told swissinfo.ch that it couldn’t provide details on the terms of the contract. Typically, in an advance purchase agreement like this, a government pays an upfront amount of money to fund further development of a vaccine in exchange for a guarantee that a certain amount of production is set aside for them. If it fails, they don’t get their money back.
According to the WHO,External link Moderna’s vaccine is one of a handful of projects in Phase Three clinical trials out of the more than 160 candidates globally. Vaccines in final trials typically have a 20% chance of failure. Although it has some 20 drugs and vaccines in development, the ten-year-old company has never had a product reach the commercial stage.
“The decision is risky and premature. Switzerland is caught up in the vaccine hunt and hype,” said Durisch. “They fear that if we don’t do it now, there won’t be anything left.”
Ten years ago, governments signed similar deals for a swine flu vaccine and eventually had to find ways to donate or get rid of them when the outbreak subsided.
Federal health authorities have indicated that they are in talks with other manufacturers. As a possible sign of more deals to come, the government announced an agreement with Swiss biotech Molecular Partners on Tuesday for access to 200,000 doses of its Mono-DARPin treatment in development against Covid-19.
Strings attached
Moderna has also attracted criticism on several fronts. The price set for the vaccine is the most expensive of the candidates and has also been highly subsidised. The US government has shelled out $955 million for Moderna’s vaccine candidate.
The CEO has also publicly said that it aims to make a profit – in contrast to Johnson & Johnson and Astra Zeneca which have said they would produce vaccines at no profit to themselves. The health industry site STAT also reported gaps in the disclosureExternal link of development costs for the Covid-19 vaccine, which is required under US federal law.
“Governments are writing blank checks without any strings attached related to price, access or transparency,” said Durisch.
Michael Altorfer, who heads the Swiss Biotech Association, argues that it is important for a company to generate a profit on its investment. “Otherwise, investors will learn the lesson and abort investments in this field,” he said.
Big pharma companies have been abandoning areas like vaccine and novel antibiotics R&D because they can’t make a return on their investment when prices are low.
He notes that as is the case with many biotech start-ups, private investors bore risks in the early stages of developing Moderna’s mRNA technology that gave the company a leg up in the race for a Covid-19 vaccine.
Switzerland also has its own stake in Moderna’s success. It has signed a deal with Lonza to manufacture the Active Pharmaceutical Ingredients (API) for the Moderna vaccine at Lonza’s operations in canton Valais.

More
Lonza ‘scaling up’ Moderna’s promising Covid-19 vaccine process
Swiss media have also reported that Moderna appears to be setting up shop in Switzerland. According to reports, the firm entered a local subsidiary into the Swiss commercial registry at the end of June. On Tuesday, the firm appointed Nicolas Chornet as its head of European operations, based in Basel.
This makes Switzerland one of the few countries in the enviable position of having both vaccine production taking place on its soil and the financial means to procure vaccines. Suerie Moon, co-director of the Global Health Centre at the Graduate Institute Geneva, says that the Swiss government should use its position to pressure Lonza to abide by WHO guidelines on allocation when producing vaccines.
“The government itself could also take a more integrated approach by procuring a large volume at once, some for Swiss use and the remainder for other countries to use, via the COVAX facility that has been strongly supported by Switzerland,” said Moon.

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative





























Join the conversation!