
Roche at-home Covid tests receive US FDA nod
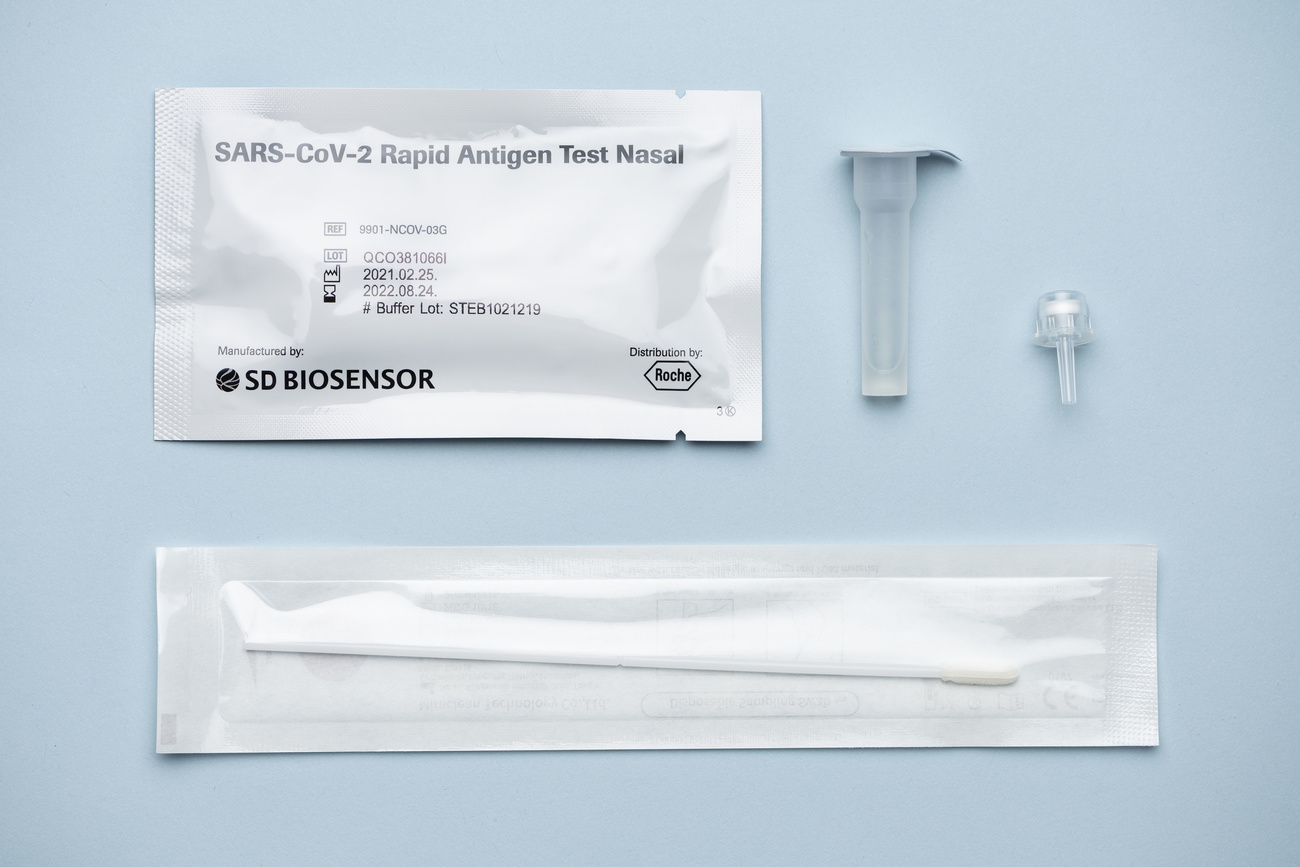
Swiss biopharma firm Roche has been granted emergency approval by US authorities to sell its at-home rapid Covid-19 test starting in January.
On Friday, Basel-based Roche announced that the US Food and Drug Administration has granted Emergency Use Authorisation for its at-home rapid test. The tests will be made available over-the-counter at pharmacies and retailers throughout the United States starting in January 2022.
“The Covid-19 pandemic continues to shed light on the critically important role that rapid self-testing plays in empowering individuals to protect their personal health and the health of their families and communities,” said Thomas Schinecker, CEO, Roche Diagnostics, in a press statementExternal link.
The Covid-19 At-Home Test uses a nasal swab sample to enable individuals to self-test at home and receive results in about 20 minutes for coronavirus and all known variants of concern, including Omicron.
The at-home tests or “self tests” have been widely used in Switzerland since this spring. For a period of time, a certain number of them were available to the population at no-cost. Studies of the accuracy of the results vary with some showing that the test detects positive cases when there is a high viral loadExternal link. One study from the University of Bern showed that the tests picked up 44% of symptom-free cases that were positive through a PCR test.
The company has a distribution agreement with SD Biosensor who has also been involved in the launch of a range of Covid tests. A key consideration for the authorisation, writes Roche, was its ability to deliver large quantities of tests and ramp up manufacturing to meet future demands. In a statement, the company said “it has the capacity to produce tens of millions of tests per month to help support the pandemic response”.

In compliance with the JTI standards
More: SWI swissinfo.ch certified by the Journalism Trust Initiative



























You can find an overview of ongoing debates with our journalists here . Please join us!
If you want to start a conversation about a topic raised in this article or want to report factual errors, email us at english@swissinfo.ch.